Aug 8, 2019
Allergan Fights to Keep Botox-Making Process Secret From Rival
, Bloomberg News
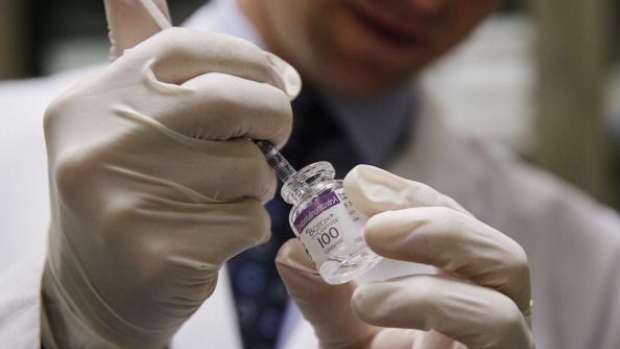
(Bloomberg) -- Allergan Plc has gone to great lengths to guard the secret of how it makes Botox, its $2 billion-a-year wrinkle treatment. Metal-enclosed workstations, security guards and private jets are part of the effort.
And now it is challenging a trade judge’s order to reveal details of the manufacturing process, which it calls one of its “most valuable and highly protected assets.”
An U.S. International Trade Commission judge told Allergan to give lawyers for Daewoong Pharmaceuticals Co. and Evolus Inc. the information as part of a legal dispute over processes to make the next generation of Botox.
Patents give their owners exclusivity for about 20 years but they have to reveal details of their invention to the public. Trade secrets can last as long as their owner keeps it away from the public. While Allergan has obtained some patents on Botox, it said it’s maintained the Botox manufacturing process confidential for three decades.
Botox, one of Allergan’s top products, is made from the deadly botulinum toxin and used to treat both wrinkles and migraines. South Korea’s Daewoong says it needs the information to defend itself against claims its product, Jeuveau, was made using a bacterial strain and process stolen from another Korean manufacturer, Medytox Inc., that’s working with Allergan.
Allergan and Medytox filed the trade-secret theft complaint with the ITC in February, asking the agency to block imports of Daewoong’s products.
Allergan said there’s no need to give Daewoong the information because the case isn’t over the actual Botox process but the next-generation treatment being developed by Medytox.
Just as important, Allergan said, is the risk to the company’s business.
“The toxin field is comprised of a small field of experts and they would be unable to unlearn information to which they gained unprecedented access,” Allergan said. “It is highly likely, if not inevitable, that experts granted access to Allergan’s technical information regarding Botox will find themselves working or consulting for Allergan’s competitors down the road.”
Rush to Market
Medytox contends Daewoong was able to beat the rush to market with Jeuveau only because a former Medytox employee gave it a copy of the bacterial strain that’s a variant of Botox. Daewoong says it developed its strain from soil in South Korea, not from Medytox.
Daewoong argues that both Allergan’s and Medytox’s processes aren’t really trade secrets. Learning how Botox is made would show that the manufacturing steps “are generally known, contained in published materials or self-evidence variations or modifications of that public information,” Daewoong said. The company said Allergan is exaggerating the threat to its business.
“Sometimes companies will file patent applications on the process and disclose it broadly, but the fine-tuning tweaks that really matter are kept secret,” said Eldora Ellison, a biotechnology lawyer with Sterne Kessler Goldstein & Fox in Washington, who isn’t involved in the case. “The manufacturing process can be very important, particularly with the quality or whether it even works or not.”
Public Domain
ITC Judge David Shaw, in his July 2 order, sided with Daewoong’s request. “Commonalities between Allergan’s process, Medytox’s process and those described in the public domain can be used as a defense to trade secret misappropriation,” he said.
The issue, which has been percolating at the Washington trade agency for a month, may force Allergan to drop the case even if it thinks it’s in the right, said Kevin Noonan, a patent lawyer who heads McDonnell Boehnen Hulbert & Berghoff’s biotechnology group in Chicago and isn’t involved in the case.
“It does put them in a really tough spot,” Noonan said.
Allergan said the ITC staff, which acts as a third party in the case on behalf of the public, originally supported disclosure of the Botox information but has since changed its position. That could boost Allergan’s request that Shaw reconsider his July 2 order, or have the issue immediately sent to the full commission for review.
The case is In the Matter of Certain Botulinum Toxin Products, Complaint No. 337-3359, U.S. International Trade Commission (Washington).
--With assistance from Cynthia Koons.
To contact the reporter on this story: Susan Decker in Washington at sdecker1@bloomberg.net
To contact the editors responsible for this story: Jon Morgan at jmorgan97@bloomberg.net, Wendy Benjaminson
©2019 Bloomberg L.P.