Jul 12, 2021
Appili Therapeutics: a David taking on the COVID-19 Goliath and other infectious diseases

- The COVID-19 pandemic might be on the downturn, but it will inevitably be followed by new viral variants along with other viruses and pathogens that are difficult to treat
- Appili Therapeutics is at the forefront of the fight against infectious diseases with a mission to find solutions to life-threatening infections
- The company has joined a global consortium of pharmaceutical, manufacturing, and distribution leaders to develop oral antiviral AVIGAN®/REEQONUS™ (Favipiravir) for COVID-19
The current pandemic has taught us is that infectious diseases have always been and will continue to be a threat to societies around the world, especially as the planet becomes increasingly globalized.
COVID-19 will inevitably be followed by COVID-20 and COVID-21. But these Coronaviruses aren't the only bugs we need to be concerned with.
A study by the National Academy of Sciences, Engineering and Medicines found that, during the past three decades, 37 new human pathogens have been identified as disease threats. An estimated 12 per cent of known human pathogens have been recognized as either emerging or re-emerging. Threats like HIV/AIDS, the Zika virus and SARS are other examples.
But it's not just viruses threatening our collective health. There are fungal infections, tropical diseases, and other pathogens out there that are either hard or nearly impossible to treat with current therapies.
“We are running a large international Phase 3 clinical trial for an oral antiviral for COVID-19.”
— Armand Balboni, MD, PhD, CEO, Appili Therapeutics Inc.
Beyond COVID19, drug-resistant bacteria and fungi continue to be a growing problem with few new solutions on the horizon. These emerging, re-emerging, and resistant diseases are undermining the antibiotics and antifungal drugs we’ve relied on for decades. For example, Candida auris, a highly drug resistant fungus that has a high mortality rate, was lurking in hospitals and nursing homes long before COVID-19 took up residence. It is a serious and growing threat.
Appili Therapeutics: a David taking on the Goliath of COVID-19 and other pathogens
It's therefore reassuring that flying under the radar is Halifax-headquartered Appili Therapeutics Inc. (TSX: APLI | OTCQX: APLIF), a biopharmaceutical company that has quietly built a portfolio of projects that have put it at the forefront of the global fight against infectious diseases.
The company was built to take on those human pathogens and help develop solutions to these life-threatening infections.
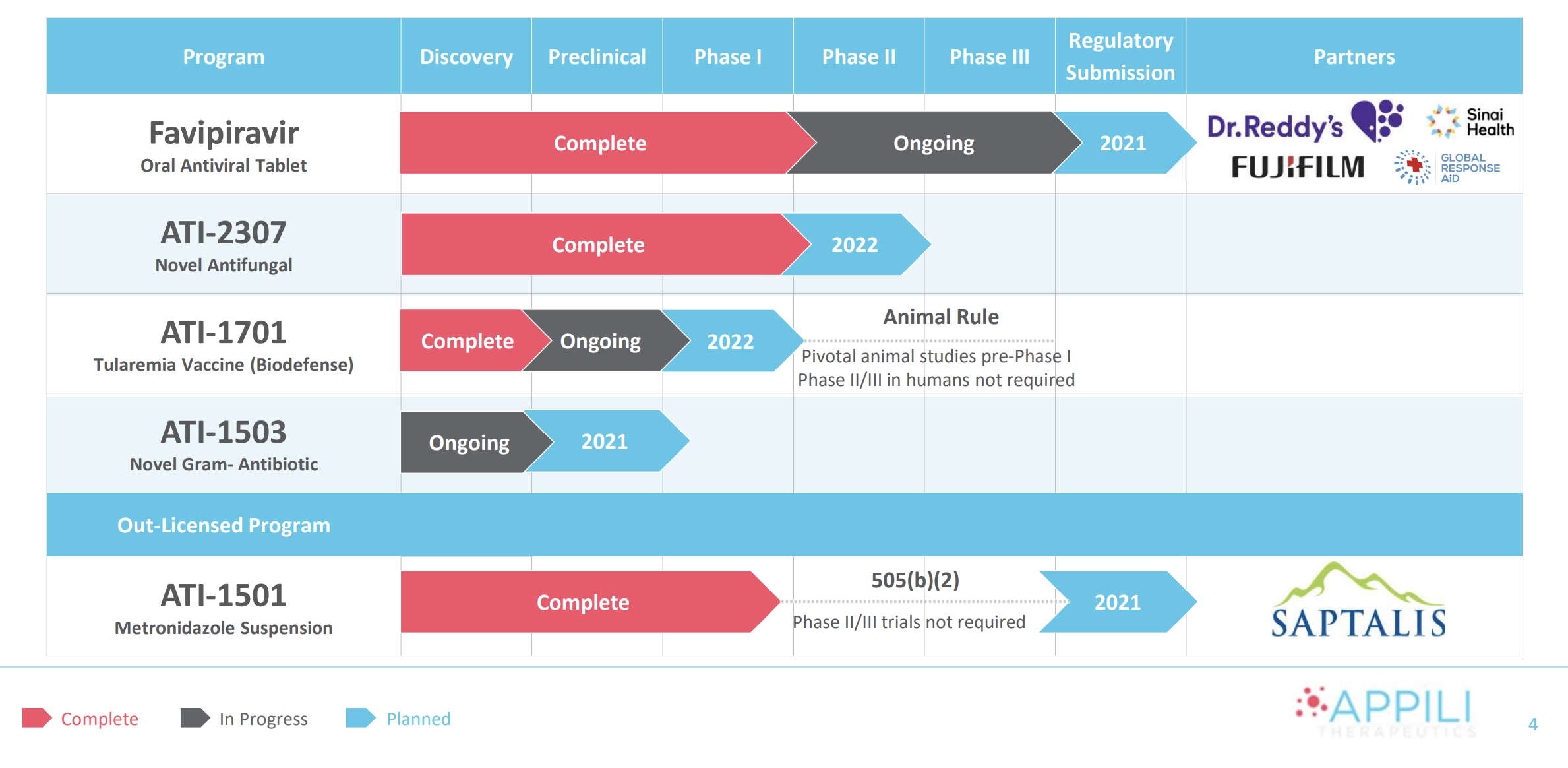
Appili’s lead program is an oral antiviral for the treatment of COVID-19. As part of a global consortium, the company is conducting a large international phase 3 clinical trial (PRESECO) evaluating the antiviral Favipiravir for the treatment of mild/moderate COVID-19 in subjects not yet hospitalized.
"There is a growing consensus that we need more tools in our toolbox to fight COVID-19. As a company, we are seeking to address this unmet need and consequently punch way above our weight in terms of what we're doing," says Appili CEO, Armand Balboni, MD, PhD.
"While Merck and Pfizer get a lot of the press, our program is in late-stage development, and we are running a large, robust phase 3 clinical trial to develop a drug for patients that currently have no oral drugs for use at home for COVID-19.”
Appili is part of the consortium with industry leaders like Dr. Reddy’s Laboratories and FUJIFILM ready to roll out product worldwide pending positive data.
The company recently expanded its trial from the U.S. into Brazil and Mexico. A promising potential early treatment for patients with mild-to-moderate COVID-19 symptoms, Favipiravir could be a game-changer. It's easy to access, can be taken at home and helps people feel better faster while staying out of hospital.
With the trial ending this summer and a report detailing the findings scheduled for September, Appili is positioned for profit sharing and royalty agreements with the consortium.
The markets are ready for an oral tablet to treat COVID-19 and one model that's a potential example is Tamiflu, a prescription flu medication, which generated $3 billion in global revenues in 2009 and over US$500 million per year in the last two years of exclusivity in the U.S.
Balboni says that “while we continue to push forward with our PRESECO study, ours is a long-term approach and I think investors need to focus on that. They may be completely focused on COVID-19 right now, but we're already looking into the future and are continuing to develop our other programs for serious fungal and bacterial infections (among others)," Balboni says.
$APLI is part of a consortium of companies, including @drreddys, Global @ResponseAid & #FUJIFILMToyama Chemical, for the worldwide development & distribution of favipiravir for the potential treatment & prevention of #COVID19. https://t.co/vEOO7LKBXM #IDTwitter #avigan
— Appili Therapeutics (@Appili) March 31, 2021
Appili's diverse projects pipeline includes critical antifungal treatment
A critical component in that project pipeline is Appili’s novel antifungal treatment, ATI-2307. Phase 1 studies have been completed on the drug and initial results show that it is safe and well-tolerated. Phase 2 studies are expected to begin in 2022.
The current standard of care of systemic fungal infections has proven to be limited by the development of drug resistance and toxicity. ATI-2307 would offer health care providers a new option in treating difficult, drug-resistant fungal infections.
Balboni notes that clinicians need new tools to fight systemic fungal infections and drugs like ATI-2307 can help fill the gap in the clinical armamentarium.
"We built a company that can do good and do well by building a diversified portfolio in a space that is otherwise neglected," explains Balboni.
Because Appili isn't bound by platform technology, the company has the flexibility to use a balanced risk approach in building its pipeline. The result is a mix of close-to-market/revenue assets that provide near-term value and cash flow coupled with cutting-edge, transformational programs that target some of the biggest infectious disease challenges faced by humanity today.
High-level partnerships, relationships help Appili fill the pharmaceutical gaps
Two other examples of Appili identifying the pharmaceutical gaps and finding solutions are the company's ATI-1701 and ATI-1503 projects.
ATI-1701 is a vaccine designed to combat weaponized Francisella tularensis, a bacteria 1,000 times more potent than anthrax and easily spread. It's thought to have been weaponized by several state actors and the development of a vaccine is a priority for Western governments. The U.S. DoD continues to support the ATI-1701 program and to date has provided millions of dollars in non-dilutive funding to Appili.
Civilian and military stockpiling of any approved vaccine could generate hundreds of millions of dollars in revenue.
By way of example, the U.S. government spent up to US$1.6 billion in 2016 alone on stockpiling 52 million units of the anti-anthrax vaccine, Biothrax.
ATI-1503 is Appili’s negamycin program. This new class of broad-spectrum antibiotics targets multi-drug resistant, Gram-negative bacteria that can cause pneumonia and other infections and are becoming increasingly drug resistant. If successful, it could be used to treat some of the top threats identified by the CDC.
As they build their project pipeline, Appili is continuing to develop partnerships and relationships in the public and private sector.
Favourable Favipiravir report in September would position Appili to meaningfully advance its development strategy
As of April, the company had raised $68 million, including $25.3 million in government assistance.
The funds have been used to advance Appili's pipeline of projects and position it to be a strategic force in the infectious disease space. Moving forward, Balboni says the company will continue to identify unmet needs and develop therapeutic solutions in the infectious disease space.
"With interim data behind us and full data on Favipiravir (PRESECO) coming this fall, we are excited to see how Favipiravir has done against COVID-19 for patients at home with mild/moderate disease," Balboni says.

"We'll continue to work on our ATI-2307 project, and I think you should see significant movement on that program as we design and get ready for clinical trials in 2022."
For Balboni and his team, it's exciting times. And very rewarding ones.
"It's exciting and a little nerve-racking trying to run a business in the middle of a pandemic, but if you're not excited about it, then I would ask ‘what are you doing in the business?’" says Balboni.
"We work hard every day to bring solutions to patients with difficult-to-treat infections; we know that we can both do good and do well. That is a powerful motivator for our employees and shareholders to be a part of this company. I am proud of what we do every day to solve these problems."
To learn more about Appili Therapeutics and their portfolio-driven, people-focused approach, visit their website here.
Make sure to follow Appili on social media for the latest company updates:
This article contains forward looking information which is based on various assumptions and subject to inherent risks, including those identified in Appili’s public disclosure available under its SEDAR profile at www.sedar.com.