Sep 28, 2022
Drug Slows Progression of Alzheimer’s in Big Trial
, Bloomberg News
(Bloomberg) -- Biogen Inc. and partner Eisai Co. surged and led other drugmakers’ shares higher amid optimism about their breakthrough trial results on Alzheimer’s disease.
The two partners said their experimental drug lecanemab significantly slowed the debilitating brain disease, making it the first medicine to blunt progression of the most common form of dementia in a large definitive study.
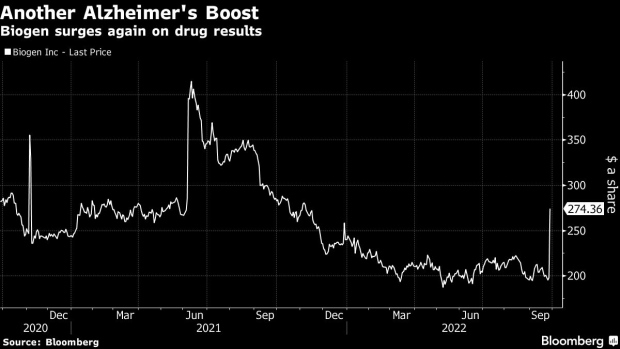
Biogen climbed as much as 43% at the US open, its biggest intraday gain since June 2021, adding more than $10 billion in market value. Eisai shares closed up 17% in Tokyo, reaching the highest level in nine months. Eli Lilly & Co. and Roche Holding AG, which are developing similar drugs, also gained.
The findings mark a major milestone for researchers who have been trying in vain for decades to stop the inexorable decline tied to the disease. How much of a difference it will make for patients and families is less clear. While it appears to unambiguously slow the disease, the medicine doesn’t restore mental capacity or totally stop its loss.
“This is clearly not a magic bullet,” said John Hardy, a professor of neuroscience at University College London. “But it looks like a definite ‘end of the beginning.”’
Read More: Biogen-Eisai Alzheimer’s Hit Doesn’t Mean Win for All
The results bolster the so-called amyloid hypothesis: a long-held but controversial theory that the buildup of amyloid, a toxic protein that clutters the brain, is one of the main causes of the disease. It therefore raises hope for other anti-amyloid drugs in development, especially medicines in final-stage trials from Roche and Lilly. Lilly, which is developing donanemab for Alzheimer’s, gained as much as 8.3%, the most intraday since December.
Roche said it was encouraged to learn of the results. Data from two key studies on its own experimental medicine, called gantenerumab, will be released at an industry conference in November. The shares rose as much as 6.5% in Swiss trading.
Challenges Ahead
Biogen and Eisai said lecanemab reduced the pace of cognitive decline in people with early disease by 27% over 18 months when compared with a placebo. The benefit came with side effects, though, including brain swelling and bleeding, though severe cases were rare.
“We think the top-line data is as strong as can be, with high statistical significance across all endpoints -- data doesn’t get much cleaner than this in biotech,” BMO Capital Markets analyst Evan Seigerman said in a note. The strength of the data reduces the chances that Medicare will deny the drug broad coverage, once it’s approved, he said.
The medicine was originally licensed from Sweden’s BioArctic AB, whose shares more than doubled on the news.
The Alzheimer’s Association welcomed the results, as did pharmaceutical and biotechnology analysts.
“We finally have what we believe to be a clean win in Alzheimer’s disease,” Evan David Seigerman, an analyst at BMO Capital Markets, wrote in a note to clients. “The top-line data are clear to us -- lecanemab slows the rate of cognitive decline.”
The trial met every goal that was set, including other measures of mental function and the ability to perform daily activities, the companies said.
The positive study isn’t the end of the challenges for Eisai and Biogen, who are collaborating on the drug that had some early controversy and will split the profits.
A previous medicine they developed together, called Aduhelm, was approved in the US in June 2021 despite contradictory trial results. While the amyloid-lowering antibody slowed the decline from Alzheimer’s modestly in one big trial, another showed no effect. Both were halted early. But Medicare refused to pay for a treatment that initially cost $56,000 a year outside of clinical trials, and the drug ended up a commercial failure.
(Adds analyst’s comment in ninth paragraph, updates shares throughout.)
©2022 Bloomberg L.P.