Jun 30, 2020
Inovio Sinks While Street Complains of Lack of Vaccine Details
, Bloomberg News
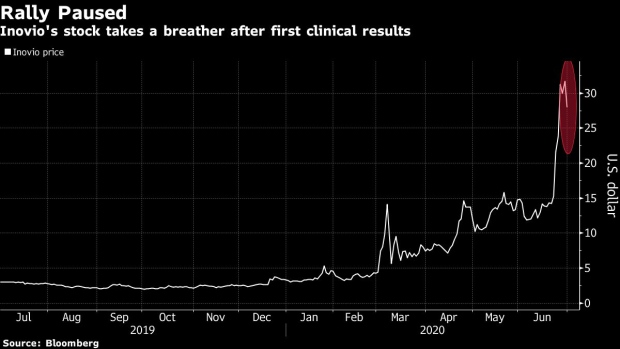
(Bloomberg) -- Inovio Pharmaceuticals Inc. sank as much as 18% on Tuesday after a first look at an experimental vaccine for Covid-19 left investors and analysts clamoring for more details.
Expectations ran high ahead of the results, which drove the stock up 860% in 2020 ahead of the data. “This preliminary data is likely to raise more questions” than answers, particularly on immune responses, Stifel analyst Stephen Willey said. He plans to wait for the full results to appear in a peer-reviewed journal before getting more constructive on the stock.
The company plans to disclose those detailed results in the coming months, Joseph Kim, the biotech company’s chief executive, said in a phone call.
For now, Inovio’s participation in Operation Warp Speed, the Trump administration’s program to speed along a vaccine for the pandemic, may “offset some of those concerns,” Willey said. Financial details of the program from all participants are scant.
Here’s what else analysts are saying:
Stifel, Stephen Willey
While the inoculation’s safety looks like a “non-issue,” for immune responses the “minimal data disclosure limits interpretation.”
Of the 36 patients for whom Inovio has immunogenicity data, 34 met one or more positive response criteria from one of the tests for antibody binding, neutralization or T-cell responses. The analyst emphasized that those 34 patients didn’t meet criteria for all three measures.
Inclusion of INO-4800 in the government program “and presumably the availability of external resources accompanying this inclusion – could serve to offset some of the investor uncertainty.”
Remains at hold after lowering the biotech company from a buy last week.
RBC, Gregory Renza
“Net-net, we are cautiously optimistic about the interim topline which showed high rate of immune responses from the study (though details on levels of responses are lacking) and encouraged by the preclinical challenge developments.”
RBC is looking for more details from the early-stage data, animal challenge studies, “as well as clarity on path forward from the FDA.”
Tuesday’s update establishes Inovio’s “continued presence in the quest for a viable vaccine.”
Reiterates at sector perform, with a speculative risk rating.
Cantor Fitzgerald, Charles Duncan
Differentiated efficacy, safety and logistics in Phase 1 study “bode well for moving forward with Covid vaccine candidate” into later stages of testing, the analyst wrote.
“Some investors may question why a comparison to convalescent serum from individuals that recovered from SARS-CoV-2 infection was not conducted. At this point, we believe there is no definitive data on what level and type of immune response is needed to protect from infection or reduce severity of disease, as well as a lack of assay standardization, and therefore we believe the comparison has clear limitations.”
Rates overweight.
©2020 Bloomberg L.P.