Sep 9, 2020
NFL season opener puts spotlight on Opko's COVID-19 testing
, Bloomberg News

The start of the National Football League season on Thursday is a milestone for Opko Health Inc., the diagnostics company that has improbably found itself at the center of the U.S.’s bid to safely resume professional sports.
Investors will be watching to see how Opko’s BioReference Laboratories handles the task of testing players and personnel as teams begin to travel and competition starts. The company has already helped Major League Soccer and the National Basketball Association resume play earlier this year.
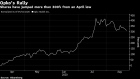
Shares of Opko, which acquired BioReference in 2015 for US$1.3 billion, surged to a two-year high in July after disclosing that it would handle testing for the NFL’s 32 teams. The stock reversed course just weeks later amid competitive concerns and a wave of false positive tests that forced the league to adjust its protocols to require repeat testing to confirm positive results in players or coaches.
“I was a little bit surprised initially” about the NFL deal, Jefferies analyst Maury Raycroft said in a telephone interview. “But to Opko’s credit they do have the infrastructure to do the testing.”
Players and team staff are tested on a daily basis, except for game day as their eligibility to play will be confirmed the day before. In the week ended Sept. 5, the NFL said 44,510 tests were administered to a total of 8,349 players and team personnel.
Contamination at a laboratory in New Jersey led to 77 false positive tests on multiple teams last month. Raycroft said BioReference could come under a cloud if it sees more problems.
The third-largest commercial lab in the country, BioReference was one of 60 testing providers the NFL contacted. A key selling point for the league was BioReference’s scale and ability to offer capacity that would not be a drain on national testing needs, Chris Halpin, the NFL’s chief strategy and growth officer, said by telephone.
“We quickly honed in on needing a managed service,” Halpin said. The league wanted a COVID-19 testing option that would enable all teams to go through the same platform.
All of the potential partners were “overall in the same ballpark” when it came to price, Halpin said. The lead up to the deal took more than a month, Halpin said. BioReference’s work with MLS and the NBA didn’t play into the decision, the company and the NFL said.
“We were having discussions with all of them essentially at the same time,” Jon Cohen, executive chairman of BioReference Laboratories, said in an interview. Neither the company nor the league shared details on the financials of the deal, though Cohen said the NFL accounts for a “very, very small” portion of the fifty to sixty thousand tests the company processes on a nightly basis.
The NFL season starts Thursday as the defending Super Bowl Champion Kansas City Chiefs host the Houston Texans.
A successful return to sports could mark a turning point for Opko investors. The company is expected to report record revenue this quarter, according to analyst estimates.
At the same time, bets against the stock are near the highest level in at least a year with about a third of the available shares sold short, data compiled by financial analytics firm S3 Partners show.
The company and its largest shareholder, billionaire Chairman and Chief Executive Officer Phillip Frost, agreed to pay US$16.5 million in June to settle a securities fraud class action alleging that the pair manipulated prices of developing health companies in which Opko had stakes in. In December 2018 Frost agreed to pay more than US$5 million to settle a complaint from the U.S. Securities and Exchange Commission about driving up the price of penny stocks.
While four of the five analysts tracked by Bloomberg recommend buying shares, modeling expectations for the company has been tough given its diverse nature. The diagnostics business has kept it in the public eye over the last few months, but many analysts have been more focused on the company’s drug portfolio.
“Six months ago the focus was primarily on the pediatric growth hormone therapeutic, and now it’s shifted and is primarily focused on COVID-19 testing,” Jefferies’s Raycroft said.