Oct 7, 2020
Regeneron seeks FDA emergency clearance for COVID-19 therapy
, Bloomberg News

Regeneron Pharmaceuticals Inc. said it has asked federal regulators to authorize its antibody treatment for COVID-19 for emergency use.
President Donald Trump received the antibody cocktail last Friday under a compassionate use program after becoming sick with the coronavirus. Trump hailed Regeneron’s treatment in a video posted on Twitter Wednesday, saying he would authorize its emergency use and make it available to Americans for free.
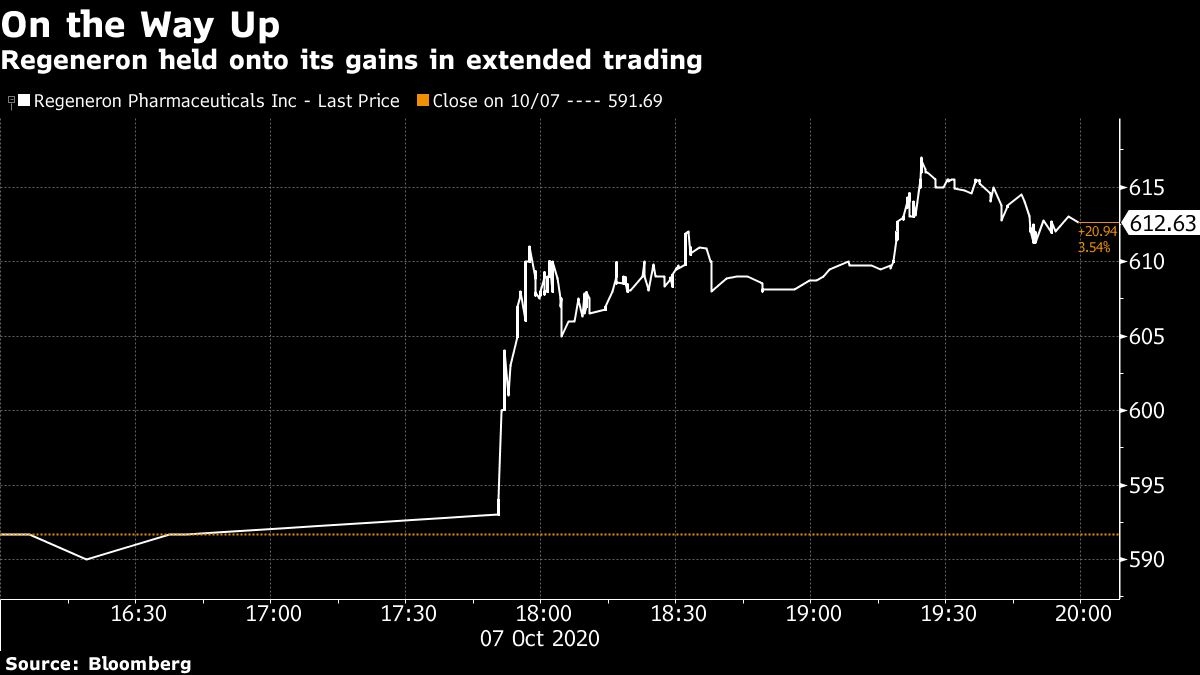
Regeneron shares gained 3.5 per cent in late trading in New York after the Trump video was posted.
The experimental therapy is one of several monoclonal antibody treatments in testing that may help treat patients with Covid-19. Late last month, Regeneron reported preliminary results from a trial showing that the cocktail could reduce virus levels in coronavirus patients outside of the hospital.
In a statement, the company said that under its agreement with the U.S. government, if an emergency authorization is granted by the U.S. Food and Drug Administration, the government would make the initial doses available to the American people at no cost and would be responsible for their distribution.
Regeneron said it currently has doses available for approximately 50,000 patients, and expects to have doses available for 300,000 patients within the next few months.
Regeneron’s move follows Eli Lilly & Co., which said earlier Wednesday it is seeking emergency authorization for its Covid antibody treatment.